Products

Diglycyl-Lysine Antibody, Clone GX41
Documentation
Main Info
- DESCRIPTION Anti-Diglycyl-lysine antibody, clone GX41
- PRESENTATION Lysophilized IgG
- STORAGE CONDITIONS Stable for 2 year at -20 ºC.
| Species Reactivity | Key Applications | Host | Format | Antibody Type |
|---|---|---|---|---|
|
|
WP, IP | Mouse | Purified | Monoclonal Antibody |
Reference for the Product:
Global analysis of lysine ubiquitination by ubiquitin remnant immunoaffinity profiling. Xu G, Paige JS, Jaffrey SR. Nat Biotechnol. 2010 Aug;28(8):868-73.
This product was cited in:
A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles. Wagner SA, Beli P, Weinert BT, Nielsen ML, Cox J, Mann M, Choudhary C. Mol Cell Proteomics. 2011 Sep;10(10):M111.013284.
Proteomic analyses reveal divergent ubiquitylation site patterns in murine tissues. Wagner SA, Beli P, Weinert BT, Schölz C, Kelstrup CD, Young C, Nielsen ML, Olsen JV, Brakebusch C, Choudhary C. Mol Cell Proteomics. 2012 Dec;11(12):1578-85.
Systems-wide analysis of ubiquitylation dynamics reveals a key role for PAF15 ubiquitylation in DNA-damage bypass. Povlsen LK, Beli P, Wagner SA, Poulsen SL, Sylvestersen KB, Poulsen JW, Nielsen ML, Bekker-Jensen S, Mailand N, Choudhary C. Nat Cell Biol. 2012 Oct;14(10):1089-98.
Irreversible inactivation of ISG15 by a viral leader protease enables alternative infection detection strategies. Swatek KN, Aumayr M, Pruneda JN, Visser LJ, Berryman S, Kueck AF, Geurink PP, Ovaa H, van Kuppeveld FJ, Tuthill TJ, Skern T, Komander D. Proc Natl Acad Sci USA. 2018 Mar 6;115(10):2371-2376
Insights into ubiquitin chain architecture using Ub-clipping. Swatek KN, Usher JL, Kueck AF, Gladkova C, Mevissen TE, Pruneda JN, Skern T, Komander D. Nature. 2019 Aug;572(7770):533-537.
Usage Statement
This product is intended for research use only and are not to be used for any other purpose, which includes but is not limited to, unauthorized commercial uses, in vitro diagnostic uses, ex vivo or in vivo therapeutic uses or any type of consumption or application to humans or animals. Due to the highly specific nature of antibodies and antigens, we cannot predict or be held responsible with respect to how Lucerna products will behave in its customers’ systems. Researchers using Lucerna products should conduct optimization studies to achieve the optimal result possible for their intended application.
Source/Purification
Monoclonal antibodies are produced by immunizing animals with a synthetic diglycine-modified histone peptide and established in hybridoma line GX41. Antibodies are purified by protein A and peptide affinity chromatography.
Quality Assurance
Each antibody batch is measured for its ability to bind to proteins containing diglycyl-lysine and to recover diglycyl-lysine-containing peptides from tryptic digests of HEK293 cells.
Licensing
Cornell University has filed a use patent on proteomic methods that use the diglycyl-lysine antibody to identify ubiquitinated proteins. All commercial users should contact Cornell University for a use license.
Specificity:
Antibody recognizes lysine residues modified by diglycine, an adduct left at sites of ubiquitination after trypsin digest.
Data:
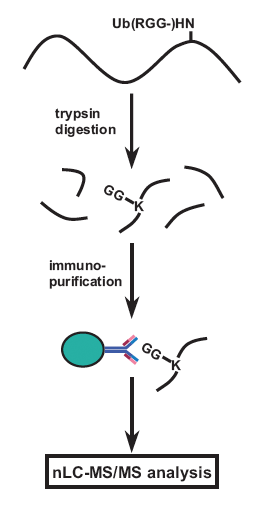
Figure 1: GX41 antibody recognizes the diglycyl-lysine moiety at ubiquitinated sites after trypsin digestion.
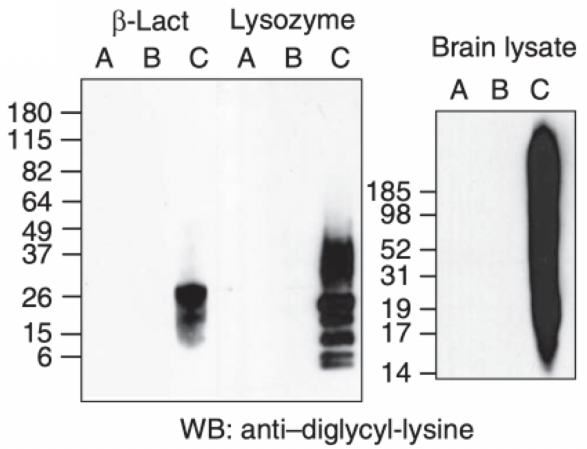
Figure 2: Specificity of anti-diglycyl-lysine (clone GX41) is verified by western blot analysis of beta-lactoglobulin, lysozyme, or rat brain lysate, in which the lysines were either unmodified (A), or modified with Boc-Gly-Gly (B) or Gly-Gly (C) adducts, respectively. As shown above, the GX41 exhibits no detectable reactivity with proteins unless the lysines are modified with Gly-Gly.
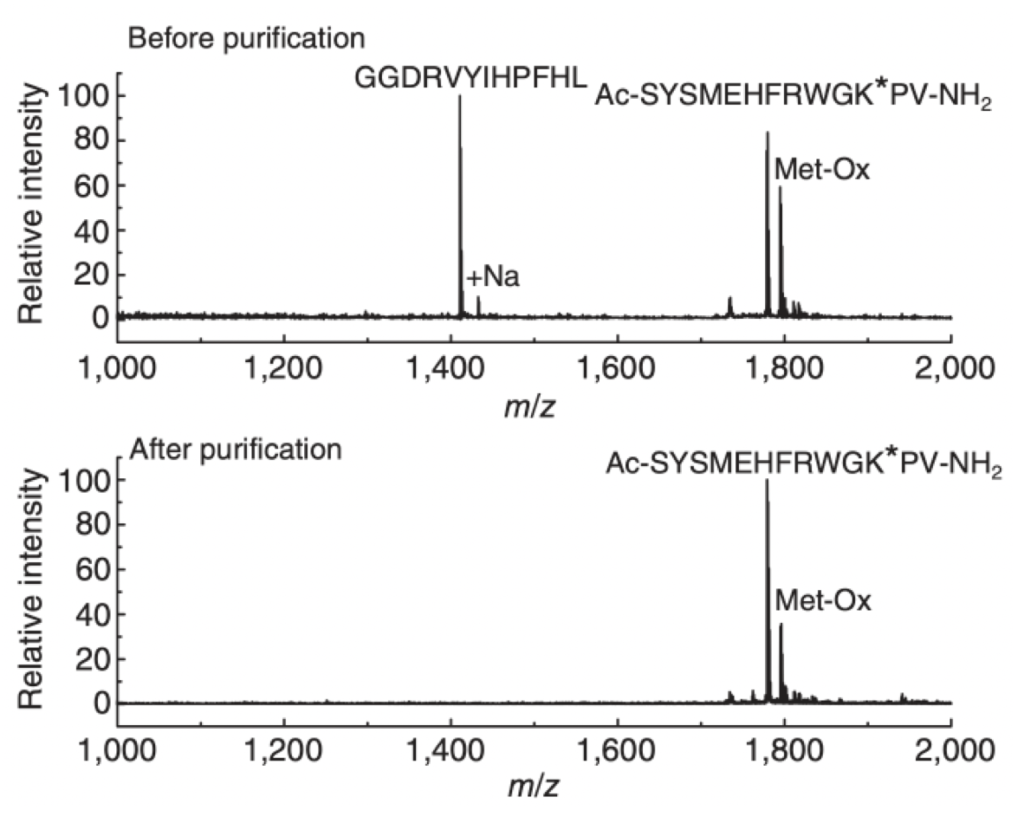
Figure 3: Equimolar amounts of the two peptides (GGDRVYIHPFHL and Ac-SYSMEHFRWGK*PV-NH2) were immunoprecipitated with immobilized GX41 antibody and analyzed by MALDI-TOF MS. K* refers to a lysine with a Gly-Gly modification. As shown above, the unmodified peptide is absent from the immunoprecipitate, while the peptide containing the Gly-Gly modification is immunoprecipitated.


